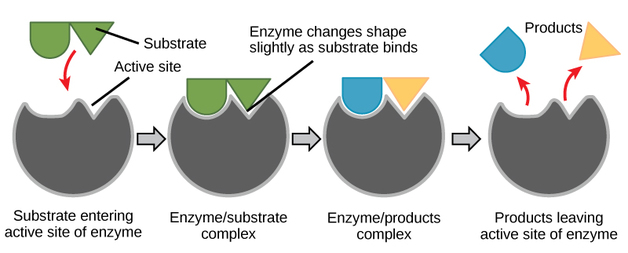
Active Sites and Enzymes
-Enzymes have an active site to which specific substrates bind
-Enzymes are globular proteins that catalyzes substrates
-The shape of the action site in the enzyme is is made to to fit in one specific type of substrate, which is why there are thousands of different enzymes with different specific-substrate
-Enzymes have an active site to which specific substrates bind
-Enzymes are globular proteins that catalyzes substrates
-The shape of the action site in the enzyme is is made to to fit in one specific type of substrate, which is why there are thousands of different enzymes with different specific-substrate
Enzyme Activity
-Enzyme catalysis involves molecular motion and the collision of substrates with the active site
-Stages of the enzyme activity
-Both enzyme and substrate are in constant random movement, which is what causes the collision between the two, most successfully if the angle makes the collision perfectly aligning the active site with the substrate
-Enzyme catalysis involves molecular motion and the collision of substrates with the active site
-Stages of the enzyme activity
- The substrate(s) attach to the enzyme's active site
- The substrate changes it's chemical composition
- The new substrate separates from the active site and leaves it vacant for the next substrate
-Both enzyme and substrate are in constant random movement, which is what causes the collision between the two, most successfully if the angle makes the collision perfectly aligning the active site with the substrate
|
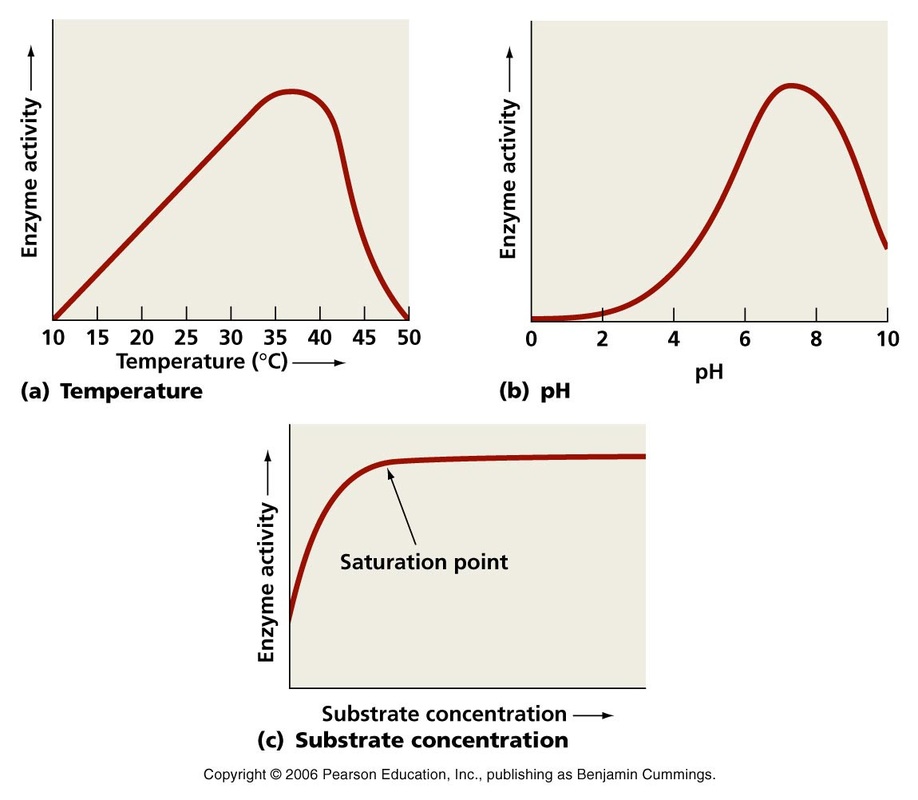
Factors Affecting Enzyme Activity
-Temperature, pH, and substrate concentration affect the rate of activity in the enzymes
|
Denaturation
-Enzymes can be denatured
-Denaturation happens when the active site changes it form and can no longer bind with the substrate
-When temperature gets too high, the enzyme bonds break and cause the active site to change form
-When an enzyme is in an environment too far from its optimum pH, the lack or abundance of hydrogen ions will also cause the enzyme to change form and denature
-Enzymes can be denatured
-Denaturation happens when the active site changes it form and can no longer bind with the substrate
-When temperature gets too high, the enzyme bonds break and cause the active site to change form
-When an enzyme is in an environment too far from its optimum pH, the lack or abundance of hydrogen ions will also cause the enzyme to change form and denature
Designing Enzyme experiments
-Design of experiment to test the effects of temperature, pH, and substrate concentration on the activity of enzymes
1. Independent variable: altering substrate concentration, temperature, and pH, including units and range
2. Dependent variable: how you are going to measure the rate of activity
3. Control variable: An independent variable that you have not altered to make sure what is the standard
Enzyme Experiments
Experimental Investigation of a factor affecting enzyme activity
-One example of enzyme experimentation is the test of different substrate concentrations of hydrogen peroxide on catalase. The independent variable would be the concentration of hydrogen peroxide, the dependent would be the amount of oxygen produces (since it is it's by-product of metabolism), and the control should be the amount of catalase.
-Design of experiment to test the effects of temperature, pH, and substrate concentration on the activity of enzymes
1. Independent variable: altering substrate concentration, temperature, and pH, including units and range
2. Dependent variable: how you are going to measure the rate of activity
3. Control variable: An independent variable that you have not altered to make sure what is the standard
Enzyme Experiments
Experimental Investigation of a factor affecting enzyme activity
-One example of enzyme experimentation is the test of different substrate concentrations of hydrogen peroxide on catalase. The independent variable would be the concentration of hydrogen peroxide, the dependent would be the amount of oxygen produces (since it is it's by-product of metabolism), and the control should be the amount of catalase.
Immobilized Enzymes
-Immobilized enzymes are widely used in industry
-Pasteur believed that enzymes could only work in living organism, the concept of vitalism
-However, the Buchner brothers discovered that by extracting the enzymes from a cell could catalyze other substances outside the living organism
-Now more than 500 enzymes have commercial uses, most of the are immobilized enzymes
-Immobilized enzymes are widely used in industry
-Pasteur believed that enzymes could only work in living organism, the concept of vitalism
-However, the Buchner brothers discovered that by extracting the enzymes from a cell could catalyze other substances outside the living organism
-Now more than 500 enzymes have commercial uses, most of the are immobilized enzymes
Lactose-free milk
-Methods of production of lactose-free milk and it's advantages
-Lactose is the sugar in milk. It's enzyme, lactase, will creak it down into glucose and galactose
-Some advantages on inverting on this enzyme are:
-Methods of production of lactose-free milk and it's advantages
-Lactose is the sugar in milk. It's enzyme, lactase, will creak it down into glucose and galactose
-Some advantages on inverting on this enzyme are:
- People who are lactose intolerant can consume lactose only on products who have used lactase to break the lactose down
- Glucose and galactose are more beneficial for food that contain milk, since they are sweeter
- For ice cream companies, glucose and galactose are more soluble than lactose, which gives a smoother texture
- Since the bacteria ferment glucose and galactose is faster, the production of yoghurt and cottage cheese is faster
TOK
Yes, knowledge should be shared where it is more applicable. Although it may not be financially beneficial, but it is an excellent way to gain even farther knowledge on the subject. We learn the most when we are able to apply knowledge in our actions, which is why lactase research should indeed be applied on Africa and Asia were there are more lactose intolerants
Yes, knowledge should be shared where it is more applicable. Although it may not be financially beneficial, but it is an excellent way to gain even farther knowledge on the subject. We learn the most when we are able to apply knowledge in our actions, which is why lactase research should indeed be applied on Africa and Asia were there are more lactose intolerants