WHY DO WE NEED WATER?
•Lubricate lungs to exhale
•Kidneys remove wastes
•Lubricate joints and allows smooth movement
-High specific heat (a lot of energy required to change water temperature) to regulate body temperature
•High heat of vaporisation (a lot of energy required to evaporate) to sweat and cool the body.
•Lubricate lungs to exhale
•Kidneys remove wastes
•Lubricate joints and allows smooth movement
-High specific heat (a lot of energy required to change water temperature) to regulate body temperature
•High heat of vaporisation (a lot of energy required to evaporate) to sweat and cool the body.
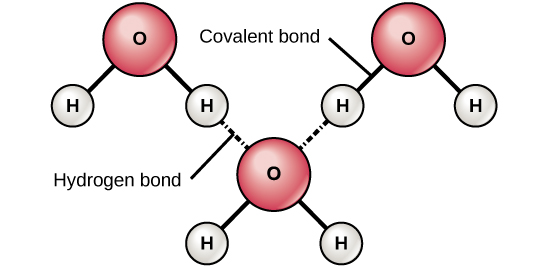
- Water molecules are formed
by a polar covalent bond between an oxygen atom and two hydrogen atoms (since hydrogen has 1 valence electrons, the two (hydrogen) valence electrons are shared with the 6 valence electrons of oxygen, reaching the 8 electrons in the outermost layers to create stability. Also, the nucleus of the oxygen atom is more attractive to the electrons than the nuclei of the hydrogen atoms (which is why its a polar covalent bond).
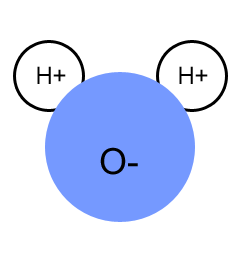
- This unbalance in electrons creates a charge, and since the water molecule is bent, the oxygen atom becomes negative and the hydrogen atoms become positive. This creates a bipolarity in the water molecule.
- Water molecules bond through hydrogen bonds. It's positive charge attracts to another atoms with a negative charge from another polar-covalent bond
|
Cohesive Properties:
Water molecules are stick to each other thanks to their hydrogen bonding. This is really useful for plants to get their water up from their roots to the rest of the plant. |
Adhesive Properties:
Water molecules can also form hydrogen bonds with other polar molecules. This is also helpful for plants to keep them moist. When they are dry, the water molecules in the air adhere to the cell walls on the leafs |
Thermal Properties:
- High specific heat: In order to increase the temperature of the water, hydrogen bonds need to be broken, and this requires a lot of energy. To cool it down, it requires to lose a lot of energy, meaning that this is beneficial for aquatic organisms because their habitat will usually stay in a stable temperature.
- High latent heat of vaporisation: Similarly, in order to vaporise water, the water molecules must separate and become a vapour molecule, which requires a lot of heat, there for, energy. Since vaporisation has a cooling effect, this is a good evaporative coolant for animals, who release the energy as sweat
- High boiling point: For the same reasons, it takes a lot of heat and energy for water to boil. Meaning that water can be liquid in a wide range of temperatures (0-100 °C)
Solvent Properties:
Water dissolves molecules by creating hydrogen bonds only with charged and polar molecules. The oxygen atom attracts to the positively charged ions, while the hydrogen atoms attract to the negatively charged ions, making them both dissolve. The water molecules form shells around the substance's molecules to prevent them from sticking together and keeping them in solution.
Water dissolves molecules by creating hydrogen bonds only with charged and polar molecules. The oxygen atom attracts to the positively charged ions, while the hydrogen atoms attract to the negatively charged ions, making them both dissolve. The water molecules form shells around the substance's molecules to prevent them from sticking together and keeping them in solution.
Hydrophilic/Hydrophobic
Hydrophilic substances are those with polar and charged molecules (to which water forms hydrogen bonds), and those that water adheres to (like cellulose)
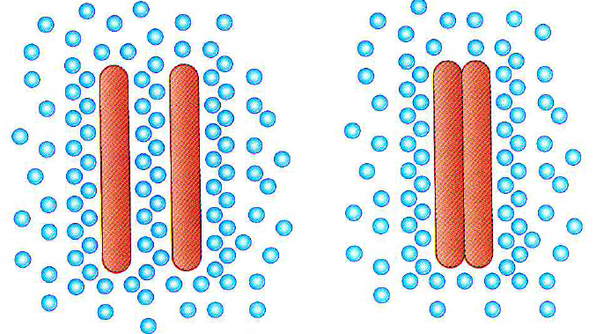
Hydrophobic substances are non-polar molecules. When these substances are submerged in water, the water molecules surround this non-polar molecule. When there are several non-polar molecules surrounded by water molecules, they attract to each other and ignore the water molecules surrounding them. These are known as hydrophobic interactions.
Hydrophilic substances are those with polar and charged molecules (to which water forms hydrogen bonds), and those that water adheres to (like cellulose)
Hydrophobic substances are non-polar molecules. When these substances are submerged in water, the water molecules surround this non-polar molecule. When there are several non-polar molecules surrounded by water molecules, they attract to each other and ignore the water molecules surrounding them. These are known as hydrophobic interactions.
Solubility of substances in our blood
- Amino acids have both negative and positive ions, which means that it is soluble in water. However, some R groups are hydrophobic. Even so, all amino acids are soluble enough to be transported through our blood.
- Glucose is freely soluble, having both negative and positive ions. With ease, it dissolves in water and transported through our blood
- Oxygen is non-polar, but because of its small size, it dissolves in water. However, it does not dissolve well and blood needs hemoglobins to transport a good amount of oxygen
- Fats are completely non-polar, and are carried by lipoprotein complexes throughout the veins
- Similarly, cholesterol is non-polar. Although it does have a small hydrophilic fraction, it is not enough to dissolve in water, there for, it has to be carried in the lipoprotein complexes with the fats
Water vs Methane
Both water and methane molecules are small and linked by single covalent bonds. However, methane is not polar, it does not have charged particles (like hydrogen and oxygen), there for, it doesn't create hydrogen bonds. Without these hydrogen bonds, it is unable to develop all the properties shown above.
Both water and methane molecules are small and linked by single covalent bonds. However, methane is not polar, it does not have charged particles (like hydrogen and oxygen), there for, it doesn't create hydrogen bonds. Without these hydrogen bonds, it is unable to develop all the properties shown above.